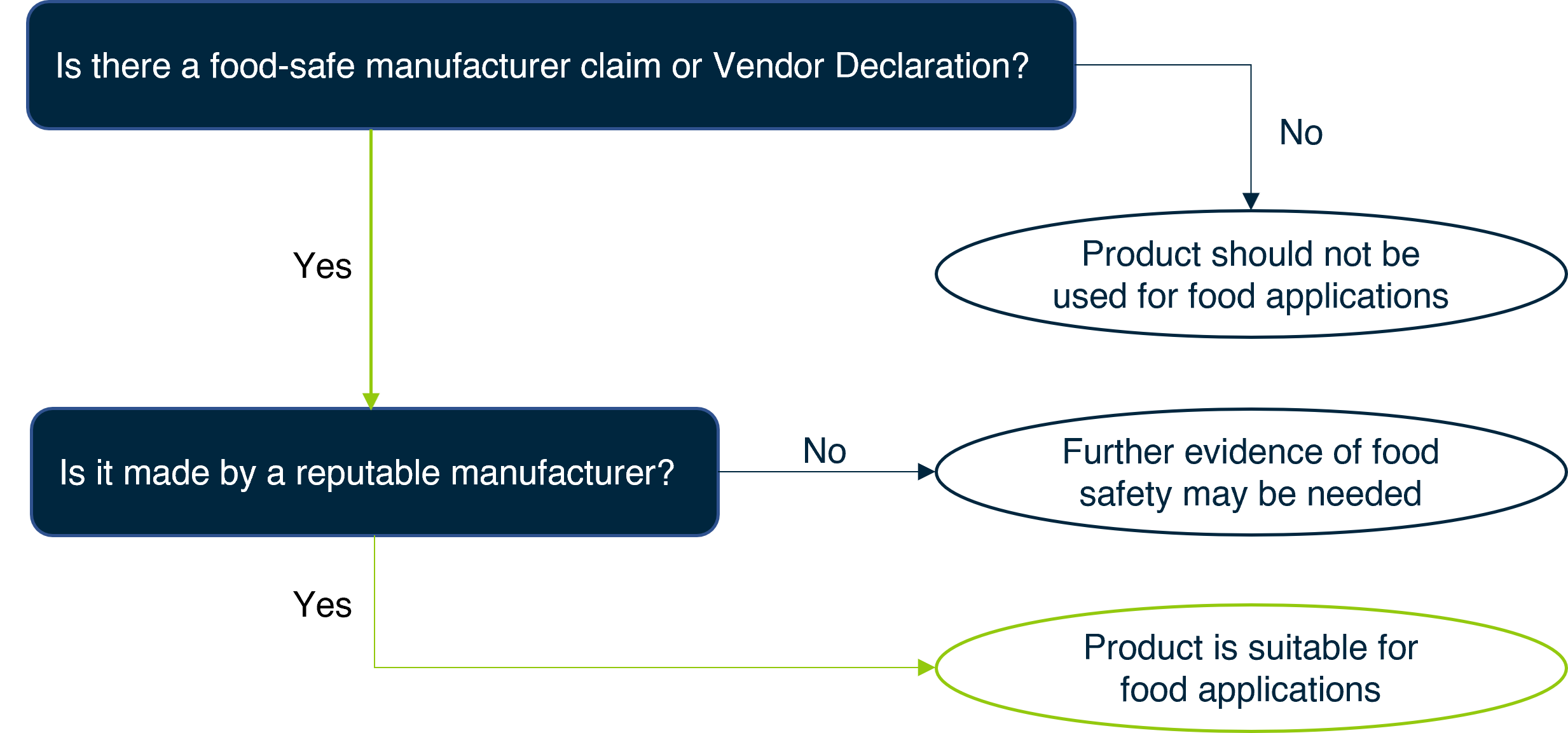
Identifying a FitForFood product
On this page:
Cleaning and sanitising products have been an essential part of food hygiene for decades. Consequently, most chemicals that are used as cleaners and sanitisers are well established.
A suggested approach for determining if a cleaner or sanitiser is suitable for food use is outlined below.
Level 1: Manufacturer claims and declarations
For most products, relying on the product claim or a reputable Vendor Declaration (if available, see below) is sufficient to be sure that the product is suitable for food contact applications. Many manufacturers have an established history and good reputation in the market, meaning that they have the expertise, knowledge and quality systems in place to deliver these products with confidence.
Manufacturers will hold evidence of the performance of their products. This can help food businesses to determine if a particular product is suitable for their requirements and is particularly important for businesses that have specific microbial requirements, such as higher-risk establishments that handle raw seafood or poultry. However, it is also important to note that how a product is used is a critical part of ensuring appropriate food safety—both to ensure adequate performance (cleaning or sanitising) and that there is no potentially harmful residue.
If a product claims to be ‘food safe’ or in some way indicates that it is suitable for use in food contact applications, it is required by law to be so. This falls under the ‘automatic guarantee’ area of Australian Consumer Law. That is, the product must match its provided description and be fit for its purpose. However, you should still be cautious if this claim is not from a reputable supplier/manufacturer.
A label claim is the quickest and most straightforward indicator that a product is suitable for food applications. Website or printed product information may also specify the intended use of the product.
For further details, contact the manufacturer. It is in no manufacturer’s interest to misrepresent its products or put unsafe products on the market—performance and reputation are good for business and customers. They should hold the necessary data or information to back up their food-use claim (see Evidence of Food Safety, below) and be happy to share it with their customers upon request.
Accord’s Guide on Cleaners and Sanitisers that may have Incidental Contact with Food includes a Vendor Declaration for a hazardous chemical substance for use in food establishments. In signing the Declaration for a product, companies are stating that the product is suitable for use in food applications.
This approach mirrors that of the Federal Department of Agriculture, Water and Environment (www.awe.gov.au) to identify chemical products that are suitable for use in export meat facilities, including chemical products for cleaning and sanitising in areas where edible food products are prepared. Vendors declare that their product, when used in accordance with the directions on the label, is fit for the specified purpose and will not contaminate animals, meat and meat products. They also provide supporting information to back this up.
Is the product manufactured and/or supplied by a reputable business? Industry association membership or a proven track record over many years of trading can be indicators of reliable businesses and quality products.
Accord Australasia is the industry association representing manufacturers and suppliers of hygiene products, including those for food applications. Accord’s member companies are committed to advancing the reputation of the industry. They contribute to work that supports and strengthens the quality, safety and environmental credentials of the entire industry.
For example, Accord first developed the Guide on Cleaners and Sanitisers that may have Incidental Contact with Food in 2005 and has updated it periodically. This is a helpful resource for the food cleaning and sanitising products industry as it assists manufacturers and distributors of cleaning and sanitising products to locate data to demonstrate that their products are suitable for food use.
Level 2: Evidence of food safety
Manufacturers need to hold evidence that their products can be safely used in food applications. And, in the case of sanitisers, their antimicrobial efficacy.
Below is a two-tiered approach to determining the safety and suitability of food contact cleaning and sanitising products
Tier 1 evidence
There are several common, approved chemicals for use in food contact applications with long histories of safe use. These are listed in the table below, including where referenced by Australian or overseas regulators or expert bodies.
Please note that the ingredients in the table below are by no means the only suitable chemicals for food use, they are simply some of the most common ones. Indeed, as responsible manufacturers are always looking for ways to innovate and further improve their formulations, this must not be seen as an exhaustive list.
If the ingredient is not listed below, some additional resources that may help identify food-safe ingredients are included as Tier 2 evidence, below.
| Ingredient | Listing | Details/link |
|---|---|---|
| Sodium hypochlorite (‘chlorine bleach’) | ANZ Food Standards Schedule 18 – Processing aids | Permitted bleaching, washing and peeling agents—various foods (S18—7) |
| DAWE •Chemicals and/or chemical compounds (hazardous substances) to be used at export registered establishments •Broad spectrum disinfectants for approved arrangements | •Hazardous substance in common use with general acceptance (see Section 4) •18-2017 - Update to Disinfectant Lists for Approved Arrangements (AA) |
|
| US FDA •Secondary direct food additive permitted in food for human consumption •Sanitising solutions – limit/conditions specified | •Chemical used in washing or to assist in the peeling of fruits and vegetables (21CFR173.315) •Component of sanitiser solution for food use (21CFR178.1010) |
|
| Hydrogen peroxide | ANZ Food Standards Code Schedule 18 – Processing aids | Permitted processing aids for water (S18—6) |
| DAWE •Chemicals and/or chemical compounds (hazardous substances) to be used at export registered establishments | •Hazardous substance in common use with general acceptance (see Section 4) |
|
| US FDA •GRAS •Secondary direct food additive permitted in food for human consumption •Sanitising solutions – limit/conditions specified | •Direct food substances affirmed as GRAS (21CFR184.1366)•Chemical used in washing or to assist in the peeling of fruits and vegetables (21CFR173.315) •Component of sanitiser solution for food use (21CFR178.1010) |
|
| Peracetic acid | US FDA •Secondary direct food additive permitted in food for human consumption – limit/conditions specified •Sanitising Solutions – limit/conditions specified | •Chemicals used in washing or to assist in the peeling of fruits and vegetables (21CFR173.315) •Component of sanitiser solution for food use (21CFR178.1010) |
| Sodium hydroxide | ANZ Food Standards Code •Schedule 15 – Food additives •Schedule 18 – Processing aids | •Food for special medical purposes (Section 13.5) •Generally permitted processing aid (S18—2) |
| DAWE •Chemicals and/or chemical compounds (hazardous substances) to be used at export registered establishments | •Hazardous substance in common use with general acceptance (see Section 4) |
|
| US FDA •GRAS •Sanitising solutions – limit/conditions specified | •Direct food substances affirmed as GRAS (21CFR184.1763) •Component of sanitiser solution for food use (21CFR178.1010) |
|
| International •General Standard for Food Additives 192-1995 / EU food additives (ANNEX III) | •INS no. 524 / E 524 |
|
| Other hydroxides: ammonium, magnesium, potassium | ANZ Food Standards Code Schedule 18 – Processing aids | Permitted bleaching, washing and peeling agents—various foods (S18—7) |
| US FDA •GRAS | •Direct food substances affirmed as GRAS (21CFR184.1139, ammonium; 21CFR184.1428, magnesium; 21CFR184.1631, potassium) |
|
| International •General Standard for Food Additives 192-1995 / EU food additives (ANNEX III) | •INS no. 527 / E 527 (ammonium), INS no. 525 / E 525 (potassium), INS no. 528 / E 528 (magnesium) |
|
| Triethanolamine | US FDA •Secondary direct food additive permitted in food for human consumption (flume water) – limit/rinsing conditions specified | •Chemicals used in washing or to assist in the peeling of fruits and vegetables. (21CFR173.315) |
| Acids | ||
| Citric acid | ANZ Food Standards Code Schedule 15 – Food additives | Various listings |
| DAWE •Chemicals and/or chemical compounds (hazardous substances) to be used at export registered establishments | •Hazardous substance in common use with general acceptance (see Section 4) |
|
| US FDA •GRAS •Sanitising solutions – limit/conditions specified | •Direct food substances affirmed as GRAS (21CFR184.1033) •Component of sanitiser solution for food use (21CFR178.1010) |
|
| International •JECFA Flavour •General Standard for Food Additives 192-1995 / EU food additives (ANNEX III) | •JEFCA no. 218 •INS no. 330 / E 330 |
|
| Lactic acid | ANZ Food Standards Code Schedule 15 – Food additives | Various listings |
| DAWE •Chemicals and/or chemical compounds (hazardous substances) to be used at export registered establishments | •Hazardous substance in common use with general acceptance (see Section 4) |
|
| US FDA •GRAS | •Direct food substances affirmed as GRAS (21CFR184.1061) |
|
| International •JECFA Flavour •General Standard for Food Additives 192-1995 / EU food additives (ANNEX III) | •JEFCA no. 930 •INS no. 270 / E 270 |
|
| Phosphoric acid | ANZ Food Standards Code •Schedule 15 – Food additives •Schedule 18 – Processing aids | •Food for special medical purposes (Section 15.5) •Generally permitted processing aids (S18—2) |
| DAWE •Chemicals and/or chemical compounds (hazardous substances) to be used at export registered establishments | •Hazardous substance in common use with general acceptance (see Section 4) |
|
| US FDA •GRAS | •Multiple Purpose GRAS Food Substance (21CFR182.1073) |
|
| International •General Standard for Food Additives 192-1995 / EU food additives (ANNEX III) | •INS no. 338 / E 338 |
|
| Surfactants | ||
| Sodium lauryl sulphate | ANZ Food Standards Code Schedule 18 – Processing aids | Generally permitted processing aids (S18—2) |
| US FDA •Food additive permitted for direct addition to food for human consumption | •Multipurpose additives (21CFR172.822) |
|
| Ethoxylated fatty alcohols | ANZ Food Standards Code Schedule 18 – Processing aids | Generally permitted processing aids (S18—2) |
| Alcohols | ||
| Ethyl alcohol (ethanol) | ANZ Food Standards Code Schedule 18 – Processing aids | Generally permitted processing aids (S18—2) |
| International •JECFA flavour | •JEFCA no. 41 |
|
| Isopropyl alcohol | ANZ Food Standards Code Schedule 18 – Processing aids | Generally permitted processing aids (S18—2, S18—3) |
| International •JECFA flavour | •JEFCA no. 277 |
|
| Quaternary ammonium compounds ‘QUATS’ (various) | DAWE •Broad Spectrum Disinfectants - Suitable for disinfection of surfaces and equipment at approved arrangements sites | •Various QUATS recognised, e.g., benzalkonium chloride for Class 3.0 (plant processing) approved arrangements |
| US FDA •Sanitising Solutions | •Component of sanitiser solution for food use (21CFR178.1010). Various QUATS recognised, e.g., n-alkyl (C12-C18) benzyldimethylammonium chlorides, n-alkyl (C12-C18) dimethyl ethylbenzyl ammonium chlorides; di-n-alkyl (C8-C10) dimethyl ammonium chlorides |
|
| Europe •Various quaternary ammonium compounds are widely used and accepted in food businesses across the EU | •For example, maximum residue levels (MRLs) of 0.1 mg/kg for didecyldimethylammonium chloride (DDAC) and benzalkonium chloride (BAC) were determined by EFSA to not pose any potential consumer health risks. |
- DAWE: Australian Federal Department of Agriculture, Water and Environment
- EFSA: European Food Safety Authority
- FDA: US Food & Drug Administration
- GRAS: Generally regarded as safe
- INS: International numbering system for food additives (and see searchable database)
- JECFA: Joint Expert Committee on Food Additives (and see searchable database)
There are red flags for the use of some substances in food-contact cleaners and sanitisers. For example, the following are not acceptable for use in export meat–registered establishments:
- antimony, arsenic, cadmium, lead, mercury, selenium
- carcinogens, mutagens, and teratogens classified as hazardous substances*
- chromic acid, formaldehyde, hydrofluoric acid, hydrofluorosilicic acid, oxalic acid, or their salts
- odorous compounds such as heavy perfumes, isomers of dichlorobenzene, pine oil, etc.
However, these substances may be permissible in cleaners and sanitisers for food contact applications if the manufacturer can demonstrate that they are fit for use and will not compromise food safety. For example, the Food Standards Code (Schedule 3, Identity and Purity, S3—4) does not ban lead, arsenic, cadmium or mercury; rather, it specifies limits of 2.0, 1.0, 1.0 and 1.0 mg/kg, respectively. Additionally, the 2021 Model Work Health and Safety Regulations specify cut-off values/concentration limits of ingredients in a mixture that would trigger the classification of the mixture as a carcinogen: > 0.1% for class 1A and 1B carcinogens and > 1.0% for class 2 carcinogens.
It is essential to remember that it is the dose of a substance, not merely its presence, that can potentially cause harm.
*The Globally Harmonised System for the classification & labelling of chemicals (GHS) classifies certain substances as hazardous carcinogens, mutagens and/or reproductive toxins (teratogens). Safe Work Australia’s Hazardous Chemical Information System can be searched to find whether an existing chemical is classified as a carcinogen, mutagen or reproductive toxin.
Tier 2 evidence
If there is insufficient certainty on the suitability of a cleaning or sanitising product based on Tier 1 evidence, more detailed product information may be needed.
This section points to additional resources that can be used to determine if a product is suitable for food use. The two key aspects to consider are product efficacy and chemical residues.
Resources that relate to the performance of sanitisers and cleaning products are listed below. Ask the product manufacturer for evidence of the antimicrobial performance of the sanitiser. (This is not relevant for cleaning products unless they also have a sanitising function.)
- Standard 1.6.1 and Schedule 27: Microbiological limits in food
Standard 1.6.1 sets out the maximum levels of microorganisms in specific foods and thereby guides manufacturers of sanitisers regarding the expected levels of hygiene to be achieved on food contact surfaces in food manufacturing and retail applications. Schedule 27 lists maximum limits for microorganisms in different foods.
- FSANZ Compendium of Microbiological Criteria for Food
The Compendium of Microbiological Criteria for Food is a compilation of process hygiene criteria that have been established for specific foods. It includes a section on environmental monitoring (for Listeria monocytogenes) via sampling of food contact surfaces.
- Therapeutic Goods Order 104 – (Standard for Disinfectants and Sanitary Products) (TGO 104), March 2019
The Therapeutic Goods Administration (TGA) regulates hard surface disinfectants including hospital, household and commercial grade disinfectants and sanitisers. All disinfectants must meet the efficacy standards prescribed in TGO 104. These include:
-
-
- the TGA disinfectant test, which determines the product’s ability to kill microorganisms
- disinfectant performance tests, which look at physical and chemical properties of the product as evidence of its stability for the duration of its shelf life.
-
‘Listed’ disinfectants include disinfectants that make specific claims in relation to killing microorganisms. These undergo a pre-market safety assessment by the TGA and are included on the Australian Register of Therapeutic Goods (ARTG). ‘Exempt’ disinfectants are disinfectants that do not make specific claims, are not included on the ARTG and their safety must be established by the manufacturer/supplier. More information can be found in TGA’s Disinfectant Claim Guide.
TGA approval of a sanitiser points to the existence of evidence concerning product efficacy. However, TGA approval does not mean that the product has been assessed for food safety. See the section on chemical residues, below, for resources to help assess the suitability of products from a residue perspective.
- Guidelines for Efficacy Evaluation of On-farm Dairy Cleansers and Sanitisers
Dairy cleansers and sanitisers for on-farm use must be registered by the Australian Pesticides and Veterinary Medicines Authority (APVMA) before being supplied or used in Australia. Registration addresses both the active cleaning/sanitising agent and the product formulation, with the APVMA assessment including an evaluation of the toxicity of the chemical as well as assessment of residue safety. The efficacy of these products can be demonstrated in three ways, with the amount of data to be provided based on the novelty of the product:
-
- reliance on a reference product (when the product is closely similar)
- bioequivalence data and/or scientific argument (when the product is similar; that is, the same active constituent/s at the same or similar concentration/s and the same or similar use pattern to a reference product)
- stand-alone data (when no similar approved product exists)
The APVMA maintains a database of approved products (search ‘sanitiser’ or ‘disinfectant’). Many of these include QUATs, chlorine-based or acidic actives. However, dairy cleansers and sanitisers should not be assumed to be approved, food-safe or fit for use in other food premises.
There are limited standards available to assess the efficacy of hard surface cleaning products. This is largely because cleaning performance is assessed visually (which is not possible for antimicrobial efficacy). Ineffective cleaning products are easily found out! Also, many of the detergents used in cleaning products have a long usage history and the concentrations and conditions required for effective product performance are well known. Regardless, companies may hold data on cleaning product efficacy.
The below standards relate to hard surface cleaning:
- ASTM G122 – 20: Standard Test Method for Evaluating the Effectiveness of Cleaning Agents and Processes
- DCC-17: Tough Greasy Kitchen Soil Test Methods A and B
The safety of chemical residues on food-contact surfaces is also an essential consideration for no-rinse food-contact products, such as leave-on sanitisers.
Residues are not a concern for rinse-off products. For example, all cleaning products require rinsing after use to remove food waste, dirt and other visible matter from the surface—sufficient rinsing will also remove chemical residues from the cleaning product. The same is true for a rinse-off sanitiser.
Most chemicals used as active ingredients in food-safe cleaners and sanitisers are well established.
Below are some additional resources from Australian or overseas regulations relating to substances for food contact. These lists may also specify residue limits resulting from their use in food manufacturing and preparation.
- Processing aids
The Food Standards Code (Standard 1.3.3) allows the use of certain substances as food processing aids. Processing aids are used during food processing to perform a technological purpose but have no technological purpose in the final food product.
Schedule 18 of the Food Standards Code lists chemicals that can be used as food processing aids if their residues in the final food product remain below specified limits. There is no specific list for sanitisers, but any chemical sanitisers that contain substances listed as ‘Generally permitted processing aids’ (Schedule 18—2), ‘Permitted processing aids for certain purposes’ (Schedule 18—3) and ‘Permitted bleaching, washing and peeling agents’ (Schedule 18—7) and for which the manufacturer’s instructions result in the residual limits being below those stated in the table could be considered as suitable for food use.
Please note that the tables often include references to ‘GMP’ (Good Manufacturing Practice) as the permitted level. This basically means that the amount of substance permitted is limited to the lowest level possible in accordance with GMP. (See Standard 1.1.2—2 for the full extract from the Code).
- Identity and Purity
The Food Standards Code describes the purity required for food additives and processing aids in Schedule 3.
There is no requirement in the Code for a cleaning or sanitising chemical that may come into incidental contact with food to be prepared to the Standard prescribed for the same chemical when intended for direct addition to food. Nonetheless, where a specification exists in Schedule 3, any product that complies with it would be considered to be food grade.
Where this is not achievable, or no specification exists, the general contaminant levels also provide a benchmark that may be useful in determining whether a chemical is suitable for indirect food contact use or if a cleaned surface on which chemical residue may be present is acceptable for food preparation.
- Chemicals and/or chemical compounds (hazardous substances) for use at export registered establishments
The Department of Agriculture, Water and Environment (DAWE, formerly the Department of Agriculture and Water Resources) provides information on the use of hazardous substances for use in export meat establishments. That is, chemical compounds for use in areas where edible food products are prepared, handled or stored. This specifically encompasses cleaning and sanitising compounds. Vendors are required to declare that their product, when used in accordance with the directions on the label, is fit for the specified purpose and will not contaminate animals, meat and meat products. The Appendix includes a list of hazardous substances that are in common use and for which a vendor declaration is not required.
- Safe Work Australia:
Safe Work Australia’s Hazardous Chemical Information System can be searched to find whether an existing chemical is classified as hazardous to human health. Please note that not all the health hazard classifications would be relevant to food contact chemicals.
From the USA:
- Generally Recognised as Safe (GRAS) food substances are generally permitted to be used in food production in accordance with GMP. The GRAS Substances (SCOGS) Database contains over 370 GRAS substances. The Substances Added to Food also lists substances with GRAS status or approved as flavour additives to food.
- Sanitisers approved for use on food-processing equipment and utensils, and on other food-contact articles, are listed in the US Code of Federal Regulations (CFR) Title 21, Food and Drugs, Chapter I, Part 178 Indirect food additives: Adjuvants, production aids, and sanitizers (i.e., 178.1005 Hydrogen peroxide solution; 178.1010 Sanitizing solutions.)
- The US Environmental Protection Agency (EPA) regulates the use of antimicrobial pesticides, including sanitisers used on agricultural products. The presence of an EPA registration number on the labelling indicates that a product may safely be used as indicated by label instructions. Whilst most are not relevant to sanitisers, tolerances for antimicrobial pesticides are published in 40 CFR 180.
- Additionally, US EPA List N includes several disinfectant products for food use. Whilst List N specifically designates products that kill SARS-CoV-2 (the virus that causes coronavirus), it also identifies products, and their active ingredients, that are suitable as rinse and no-rinse food-contact disinfectants.
International
- The Joint Expert Committee on Food Additives (JECFA) is an international expert scientific committee that is administered jointly by the Food and Agriculture Organization of the United Nations (FAO) and the World Health Organization (WHO). The JECFA Food Additives and Flavourings databases provide the specifications for flavourings evaluated by JECFA.
- The CODEX Alimentarius General Standard for Food Additives 192-1995 lists food additives that are recognised as suitable for use in foods (according to the conditions specified in the Standard). These food additives have been assigned an acceptable daily intake (ADI) or determined to be safe by JECFA based on other criteria. While it does not explicitly address cleaning and sanitising residues, the listing of a chemical substance in the CODEX STAN 192-1995 indicates that it is likely to be suitable for food use subject in accordance with any maximum levels. The CODEX can be searched for approved food additives via the online database.
Level 3: Risk assessment for new chemistry
If the product uses a chemical that has not previously been used in food contact applications and is not listed in any of the references above, full risk analysis documentation would be needed to demonstrate that the product is food-safe. This would need to consider:
- the efficacy of the product in performing its cleaning and/or sanitising function
- the toxicological profile of the chemical ingredients (hazard)
- levels of exposure that are likely to arise from residues, if present (dose).
The product manufacturer should be able to provide this evidence in support of their food-safe claim.